 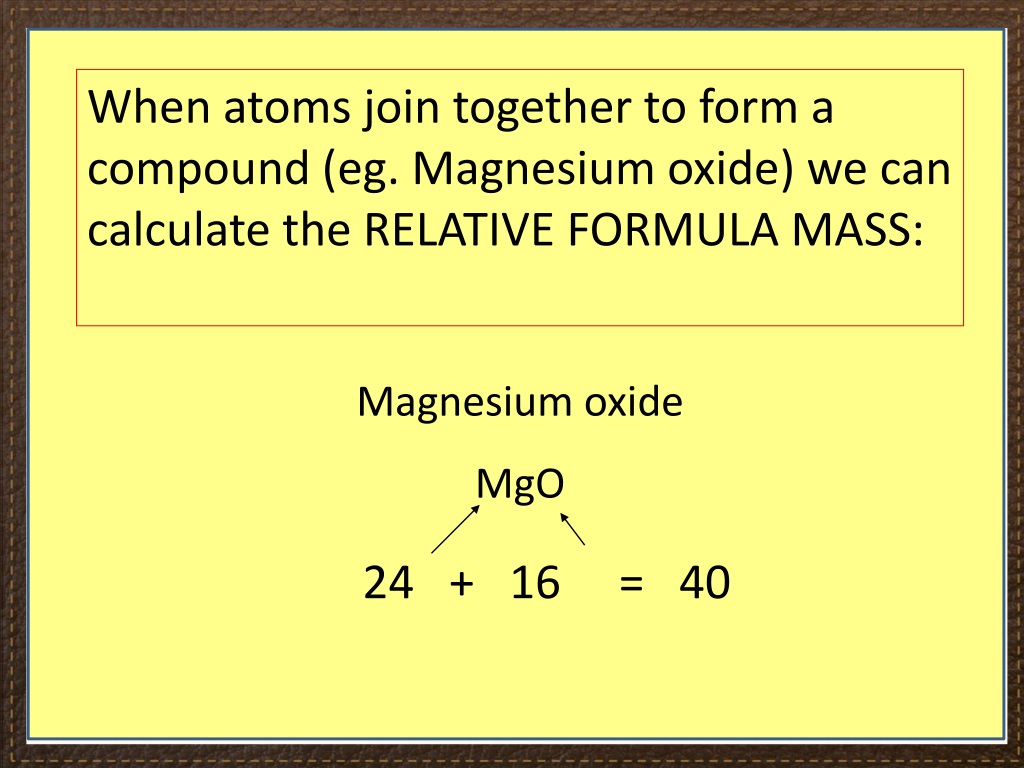
When you have a solution, click here to check your answer. If you find one, good! If you find both, excellent! There are actually two different ways of joining these up to make two different molecules. On a piece of paper, draw the electronic structures of 2 carbon atoms, 6 hydrogens and an oxygen atom, and find a way to bond them together so that there are shared electrons between each atom and its neighbour(s), and there are no unpaired electrons left over. Look carefully at this, and make sure you can see where all the electrons are coming from, and how they produce a whole lot of covalent bonds. If you have 2 carbon atoms, 5 hydrogens and a fluorine atom, there is only one way you can fit them together so that all the electrons are paired, and all the atoms are held together by shared pairs of electrons. I have chosen this because we have already got all the dots-and-crosses diagrams for C, H and F further up the page. Carbon has 4 unpaired electrons, and so can form 4 covalent bonds with hydrogen atoms. This doesn't add anything much to the discussion, but it is a frequently discussed molecule, and I include it for completeness. That maximises the amount of energy released and so makes the molecules as energetically stable as possible. They combine with enough hydrogen atoms so that all the unpaired electrons are converted into covalent bonds. That means that each of these can form more than one covalent bond. In both oxygen and nitrogen, there is more than one unpaired electron. The molecule formed has no unpaired electrons for the process to go any further. Why do they go around in pairs rather than some other number? Because each atom only has one unpaired electron which it can share with another one. Why? Because the formation of the bond between the atoms releases energy, and the molecule is energetically more stable than individual atoms. Just like hydrogen, fluorine consists of molecules made up of two atoms rather than going around as single atoms. To be on the safe side, just use dots for one atom, and crosses for the other in pencil or whatever blue or black pen you use for writing everything else.Īgain, an electron pair is shared - there was a single electron on the original hydrogen atom and also one on the fluorine atom. It doesn't matter as long as it is clear.īut be careful! Some examiners don't like you to use colour in your answers. You can use differently coloured crosses for the two atoms or you can use differently coloured dots for the two atoms. You can use dots for one atom and crosses for the other (as here). It is usual to show the electrons from the two atoms differently so that you can tell which came from which atom. Note: Diagrams like this are often described as "dots-and-crosses" diagrams. 
It is these two attractions to the same pair of electrons that acts as the "glue" holding the atoms together. So what is actually holding the atoms together? The nucleus of the left-hand hydrogen atom is strongly attracted to the electron pair the nucleus of the right-hand hydrogen atom is also attracted to the same electron pair. The electrons from the two hydrogen atoms come together to make a pair, and it is this pair of shared electrons between the two nuclei that forms the covalent bond. That is illustrated in the diagram:īy losing that amount of energy when they combine, the hydrogen molecule (with the two atoms joined together) is energetically more stable than the separated atoms.Ītoms combine together make a molecule because energy is released and the molecule is energetically more stable than its separate atoms.Ī simple covalent bond in the hydrogen molecule, H 2 So breaking bonds costs energy, and making bonds releases energy. If you allowed the hydrogen atoms to join up again, because you are simply reversing what you just did, you will get that same amount of energy out again. If you wanted to break the bond to give two separate hydrogen atoms again, it would cost you quite a lot of energy. 
Suppose you had two hydrogen atoms joined strongly together by some means. I am assuming that you can work out electronic structures of simple atoms and have access to a Periodic Table such as the one you can download from this site. This page introduces the way atoms can bond together by sharing electrons in single covalent bonds, and the various ways you can show covalent bonds on paper. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
